Student Resources
Definitions
Definitions
- Atomic Number
-
The number of protons contained within the atomic nucleus. Given the symbol Z.
- Cation
-
A positively charged ion. Cations have more protons than electrons.
- Conventional Atomic Weight
-
A single reference number for atomic weight when the sample is from an unspecified origin and unknown isotopic composition.
- Electron
-
Negatively charged subatomic particles surrounding the nucleus of an atom.
- Half-life
-
The amount of time it takes for half of a sample of a radioactive isotope to undergo a nuclear decay event.
- Isotopes
-
Atoms of the same element that have different numbers of neutrons.
- IUPAC
-
The International Union of Pure and Applied Chemistry (IUPAC) is the world authority on chemical nomenclature and terminology, including the naming of new elements in the periodic table; on standardized methods for measurement; and on atomic weights, and many other critically-evaluated data.
- Joint Working Party
-
The Joint Working Party includes experts from IUPAC and IUPAP. IUPAC is the International Union of Pure and Applied Chemistry; IUPAP is the International Union of Pure and Applied Physics.
- Long-Lived Radioisotope
-
A long-lived radioisotope is a radioactive isotope with a very long half-life. The atomic mass of a long-lived radioisotope is used in the calculation of the standard atomic weight of an element if its isotopic abundance is large enough to contribute.
- Mass Number
-
Total number of heavy particles (protons and neutrons jointly called nucleons) in the atomic nucleus. Also called nucleon number. Given the symbol A, or, in mass spectrometry, the symbol m.
- Mass Spectrometry
-
A technique for measuring relative molecular mass by accelerating charged particles (ions) by an electric field and then passing the ions through a magnetic field where they are separated according to their mass to charge ratio.
- Molecular Weight
-
Ratio of the mass of a molecule to the unified atomic mass unit. An atomic mass unit is defined as 1/12 of the mass of a 12C atom in its ground state. Therefore, molecular weight is the ratio of the mass of a molecule on a scale in which the mass of a 12C atom is exactly 12.
It is important not to confuse molecular weight with molar mass. Molecular weight is the mass of one molecule and is expressed in atomic mass units. Contrarily, molar mass is the mass of one mole of a substance and is expressed in g/mol. - Neutrons
-
Neutrally charged subatomic particles contained within the nucleus of an atom. Neutrons have about the same mass as protons and a much greater mass than electrons.
- Nucleon
-
A nuclear particle, such as a neutron or a proton.
- Nucleus
-
The positively charged central portion of an atom, excluding the electrons.
- Nuclide
-
A species of atom, characterized by its mass number, atomic number and nuclear energy state, provided that the mean life in that state is long enough to be observable.
- δ18O
-
This is a way that the isotope ratio of 18O/16O can be expressed. It is calculated from 18O and 16O values (taken from relative intensities of oxygen containing compounds determined through mass spectrometry) in a sample in comparison to a standard reference ratio called Vienna Standard Mean Ocean Water (VSMOW) with the following equation:
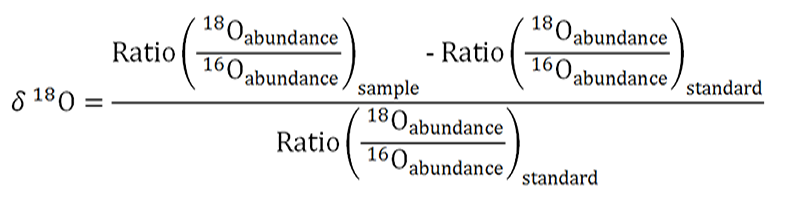
- Protons
-
Subatomic particle with a charge of +1 contained within the nucleus of an atom. Protons have about the same mass as neutrons and a much greater mass than electrons. The number of protons determines the identity of the element.
- Radiogenic
-
Refers to a nuclide created in the process of a radioactive decay.
- Radioisotope
-
A radioactive isotope of a specified element.
- Relative Atomic Mass
-
Ratio of the mass of a atom to the unified atomic mass unit. An atomic mass unit is defined as 1/12 of the mass of a 12C atom in its ground state. Therefore, relative atomic weight is the ratio of the mass of a atom on a scale in which the mass of a 12C atom is exactly 12. Given the symbol Ar.
- Stable Isotope
-
An isotope for which no radioactive decay has been experimentally detected. Stable isotopes do not undergo nuclear changes.
- Vienna Standard Mean Ocean Water
-
Often abbreviated to VSMOW, this internationally distributed stable hydrogen and oxygen isotopic reference material serves as an anchor for the stable hydrogen and oxygen isotope scales. Thus, the δ2H and δ18O values of VSMOW (Vienna Standard Mean Ocean Water) are zero.
No tooltip loaded.